All notable changes to this project will be documented on this page
RELEASE NOTES – MICROPATH 3.1.17 JUNE 25, 2021
GENERAL FUNCTIONALITY
- TM#172247 – Suppress Labels per Vial Definition
LabelCount on a ClinicalVialDefinition can now be set to “0”. Entering that ClinicalVialDefinition on a Specimen:Container prevents Specimen labels from being printed on Inlab.
RELEASE NOTES – MICROPATH 3.1.16 APRIL 2, 2021
GENERAL FUNCTIONALITY
- TM#170959 – Interface Log Table Viewer – Slow Loading
Improved performance loading records from the Interface Log table. - TM#170922 – Audit Window Enhancements
Added capability to view full audit data for truncated values and replaced ObjectIDs with corresponding value. - TM#171433 – e.Query – Rebuild a View
Added ability to rebuild e.Query Data Models, if necessary, due database structure changes. - TM#172212 – e.Query – DateTime field filters (preference)
Added option to Site Wide Reserved ManagementReports preference to allow including time, as well as date to queries that make use of Date + Time fields. e.g. Case Creation Date - TM#170922 – Audit Window Enhancements
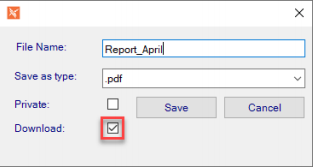
Added capability to view full audit data for truncated values and replaced ObjectIDs with corresponding value. - TM#170276 – Isolated Storage – Default Download Checkbox (preference)
Preference added to check the Download checkbox by default when saving files to the Isolated Storage Area.
RELEASE NOTES – MICROPATH 3.1.15 DECEMBER 31, 2020
GENERAL FUNCTIONALITY
- TM#169809 – Billing \ Insurance Entry Enhancements
The following enhancements have been made to the billing and insurance windows.- Standardized naming of DOB fields
- Standardized field order in windows
- Added picklist capability for: Billing.GuarantorRelation, Billing.MaritalStatus, Insurance.BenAssign, Insurance.Priority, and Insurance.SubscriberRelation
- Set Billing.SSN validation
- TM#170059 – e.Query – picklist select all (*) deselect others
Management Report pick lists now function where default of ALL (*) is now deselected when other choices are selected. - TM#170212 – Alert rules -Error Processing (preference)
Corrected issue to ensure that all alert rules are processed despite one failing due to an error. Also added preference to display notice of alert failures in Result Manager tree, so they can be corrected. - TM#170596 – Interface Map – Configurable Pick Lists (preference)
In an effort to simplify maintenance of the Interface Map library, the capability to hook up customized pick lists to the InternalCode, ExternalCode and Description fields has been implemented.
RELEASE NOTES – MICROPATH 3.1.14 NOVEMBER 9, 2020
GENERAL FUNCTIONALITY
- TM#168017 – Logo Missing- Static image handling
Images embedded to avoid access issue over networks.
INTERNAL / MINOR ENHANCEMENTS / CORRECTIONS
- TM#169873 – Edit Patient screen is erroring in V3.1.14
- TM#168921 – WIS Loader PlugIn – New Plug In e.fax Integration
- TM#169259 – e.Monitor Ellipsis in Automated Task window gives memory error
- TM#169218 – Management Report Argument issue
- TM#169778 – e.Monitor Show Backbone processes only when logged in as Admin ‘0’ account. – not complete
RELEASE NOTES – MICROPATH 3.1.13 – OCTOBER 13, 2020
NEW FUNCTIONALITY OR GENERAL CORRECTIONS
- TM#167238 – Test Status out sync with Reports
Corrected issue where Result Status could be set final without a report. - TM#168080 – PlugInFax – Implement Max Size of Fax Transaction
Ability to control the maximum size of Fax transactions for large volume faxing. - TM#167884 – PlugIn – FAX STATUS – Optimize Interfax Fax Status Updates
Improve the update status of Fax transmissions.
E.QUERY / REPORT DESIGNERS
- TM#167098 – Isolated Storage – Add Download Directory
Ability to specify private file storage locations when saving Management Reports. - TM#159663 – e.query (Data Analysis)
New utility for e.Query users to generate adhoc data collection. - TM#168279 – e.Query – Implement Display of XML data
Added ability to display XML data on management reports.
MicroPath 3.1.13 released 10/9/2020
RELEASE NOTES – MICROPATH 3.1.12 JUNE 26, 2020
NEW FUNCTIONALITY OR GENERAL CORRECTIONS
- TM#164766 – e.Monitor – Add Capability to Start and Stop Processes
The ability to stop/start/reset BackBone processes from within e.Monitor has been added. Also, processes can be temporarily hidden from view.
E.QUERY / REPORT DESIGNERS
- TM#164785 – e.Query – XML Parse Functions AAOE
Added e.Query AAOEPluck function which simplifies extracting AAOE responses. - TM#165575 – e.Query – Sync default orientation for text fields
Template for e.Query report generated fields modified so that field text alignments are set to Left Middle, which corresponds to text alignments on additionally added fields. - TM#165189 – e.Query – Added wpDateDiff function
New query function “wpDateDiff” added that calculates PatientDOB to Visit_VisitDate; includes days., months, etc. - TM#165713 – Designers – Expose Section properties on Sub Reports
Section properties available on sub reports allowing ‘Repeat’ to be set per sub report. - TM#165815 – e.Query – add NOT CONTAINS operator
Filter operator of “NOT CONTAINS” has been added to the Query Designer. - TM#165954 – e.Query – typo in function category list
Corrected - TM#166382 – e.Query – GetLibraryField function
New query function that enables extracting any field based on a library pointer ObjectId. This feature is useful when a view does not include joins to certain library tables. All library tables in a given application are supported as well as physician and user tables. - TM#166343 – RDS\C1 Report Rendering Performance
Improved report generation performance when running in an RDS environment.
MicroPath 3.1.12 released 6/26/2020
RELEASE NOTES – MICROPATH 3.1.11 MARCH 27, 2020
NEW FUNCTIONALITY OR GENERAL CORRECTIONS
- TM#163528 – Isolated Storage – Implement Private Directories
Added capability to store files for hosted clients in private, user specific sub-directories of “IsolatedStorage” directory. A check box labeled “Private” added to both the Open and Save Isolated Storage Dialog windows. - TM#163923 – Enable Suppressing On Demand prompt
The capability to suppress the on-demand prompt during order selection has been added. - TM#164590 – Audit – View Deleted button not enabled
Corrected - TM#163056 – e.Monitor enhancements including Hide, Show, Remove
Added ability to temporarily hide or remove processes from displaying in the e.Monitor Process window. - TM#164214 – Implement EMR account set up
Ability to create EMR accounts has been added to eliminate need for Outreach Manager. - TM#162943 – In EMR Activity grid, display full HL7 message in popup window.
Extremely large messages are now fully supported and displayed.
E.QUERY / REPORT DESIGNERS
- TM#163271 – Designers – Enhance Group Editor
Edit Groups menu option has been added to Other menu in all designers, allowing report groups to be added, edited for properties, or removed. - TM#163572 – e.Query – XML Parse Functions
Added capability in e.Query to select from XML data and return specified values, such as extracting the value of an AAOE response to a given question. - TM#163696 – e.Query – Add Capability to edit functions
Added ability to edit applied functions on reports, instead of removing and rebuilding.